Introduction
In 2020, a clinical trial published results that most of the Alzheimer's community overlooked—yet it showed some of the strongest treatment effects ever reported in the disease. While the world was focused on the disappointing results from anti-amyloid drug trials, the AMBAR study (Alzheimer's Management By Albumin Replacement) quietly demonstrated that plasma exchange with albumin replacement could slow cognitive and functional decline by 52-71% across multiple outcome measures.
To put this in perspective: the recently FDA-approved monoclonal antibody therapies show effect sizes ranging from 26-40% on similar measures, while carrying a 12-40% risk of brain swelling or bleeding. AMBAR showed larger effects with essentially no such risk.
So why haven't you heard about it? That's a complicated story involving regulatory pathways, industry dynamics, and the challenge of implementing a procedural intervention in a field accustomed to drug therapies. But the science speaks for itself, and it's worth understanding.
This post will walk through what the AMBAR study actually did, what it found, and what it means for people living with Alzheimer's disease. I'll present the data objectively, acknowledge the limitations, and explain why this approach warrants serious attention.
What is Plasma Exchange?
Before examining the study results, it's important to understand what plasma exchange is.
Think of your blood as having two main components: the cells (red blood cells, white blood cells, platelets) and the liquid they float in, called plasma. Plasma is mostly water, but it also contains proteins, antibodies, clotting factors, and—relevant to Alzheimer's disease—various proteins, including albumin and amyloid-beta.
Plasma exchange (also called plasmapheresis or therapeutic plasma exchange/TPE) is a medical procedure that removes your plasma and replaces it with fresh fluid. It's somewhat similar to dialysis, but instead of filtering waste products from blood, it removes and replaces the entire liquid component. It's essentially an "oil change" for the body.
The procedure works by drawing blood from a vein, separating the plasma from the blood cells using a specialized machine, mixing the blood cells with fresh replacement fluid — in the case of AMBAR, albumin — and returning the reconstituted blood to the patient. Each session takes 2–3 hours and is performed on an outpatient basis.
Why Albumin Replacement?
Albumin is the most abundant protein in human plasma — about 60% of total plasma protein — and it performs a remarkable range of functions. It transports hormones, vitamins, and drugs throughout the body. It acts as an antioxidant, protecting against oxidative damage. It binds and helps remove toxins. It modulates immune responses. And critically for Alzheimer's disease, it can bind amyloid-beta protein. In patients with Alzheimer's, albumin becomes highly oxidized and glycated — damaged by sugar molecules — losing much of its protective capacity. Replacing old, damaged plasma with fresh therapeutic albumin could theoretically restore antioxidant function, remove toxic proteins, reduce inflammation, and improve vascular health simultaneously.
The Original Hypothesis: The Peripheral Sink
The initial rationale for testing plasma exchange in Alzheimer's was based on what's called the "peripheral sink hypothesis." The idea is straightforward: amyloid-beta protein exists in equilibrium between the brain and the bloodstream. If you remove amyloid-beta from the blood, you create a concentration gradient that could pull amyloid-beta out of the brain and cerebrospinal fluid.
Think of it like opening a window in a smoky room—the smoke (amyloid-beta) will drift toward the lower concentration (the blood) from the higher concentration (the brain).
By repeatedly removing plasma containing amyloid-beta and replacing it with fresh albumin that can bind even more amyloid-beta, you might be able to gradually reduce brain amyloid burden.
That was the theory. As we'll see — and as the broader blood revolution research has since confirmed — the actual mechanism appears to be more complex and potentially more interesting.
The AMBAR Protocol: Two Phases
The AMBAR study used a specific treatment protocol with two distinct phases:
The intensive phase ran for the first six weeks, with weekly conventional plasma exchange sessions. Each session exchanged one plasma volume — calculated at 35–45 mL/kg based on patient characteristics — with replacement using 5% albumin solution, delivered via peripheral vein or central venous catheter.
The maintenance phase continued monthly for the remaining 12 months, using lower-volume plasma exchange with 20% albumin solution, alternating in some arms with intravenous immunoglobulin (IVIG) to counteract decreases in endogenous immunoglobulins from repeated plasma removal. The 2025 Argentina real-world study used a similar protocol, achieving an average of 88.2% plasma volume exchange during intensive sessions and 49.8% during maintenance — all via peripheral access, no central lines required.
The Study Design: Why This Was Serious Science
The AMBAR trial was a phase 2b/3, multicenter, randomized, blinded, placebo-controlled clinical trial. Let me break down what each of those terms means and why they matter.
Phase 2b/3: This is late-stage clinical trial work, designed to definitively test whether a treatment works and is safe enough for widespread use.
Multicenter: The study enrolled patients at 41 sites—19 in Spain and 22 in the United States. This geographic diversity helps ensure that results aren't specific to a single population or medical center.
Randomized: Patients were randomly assigned to treatment groups using a 1:1:1:1 scheme. This prevents selection bias and ensures groups are comparable at baseline.
Blinded: Patients, caregivers, and the researchers evaluating outcomes didn't know who was receiving real treatment versus placebo. This is crucial for preventing bias in subjective assessments.
Placebo-controlled: The control group underwent "sham" plasma exchange—they were connected to equipment that looked and sounded like it was performing the procedure, but no actual fluid was removed or replaced. This sophisticated placebo helps control for the psychological effects of receiving an intensive medical intervention.
The Numbers
- Screened: 496 patients evaluated for eligibility
- Randomized: 347 patients enrolled
- Treated: 322 patients actually received treatment (25 dropped out before starting)
- Completed: 232 patients finished the full 14-month protocol
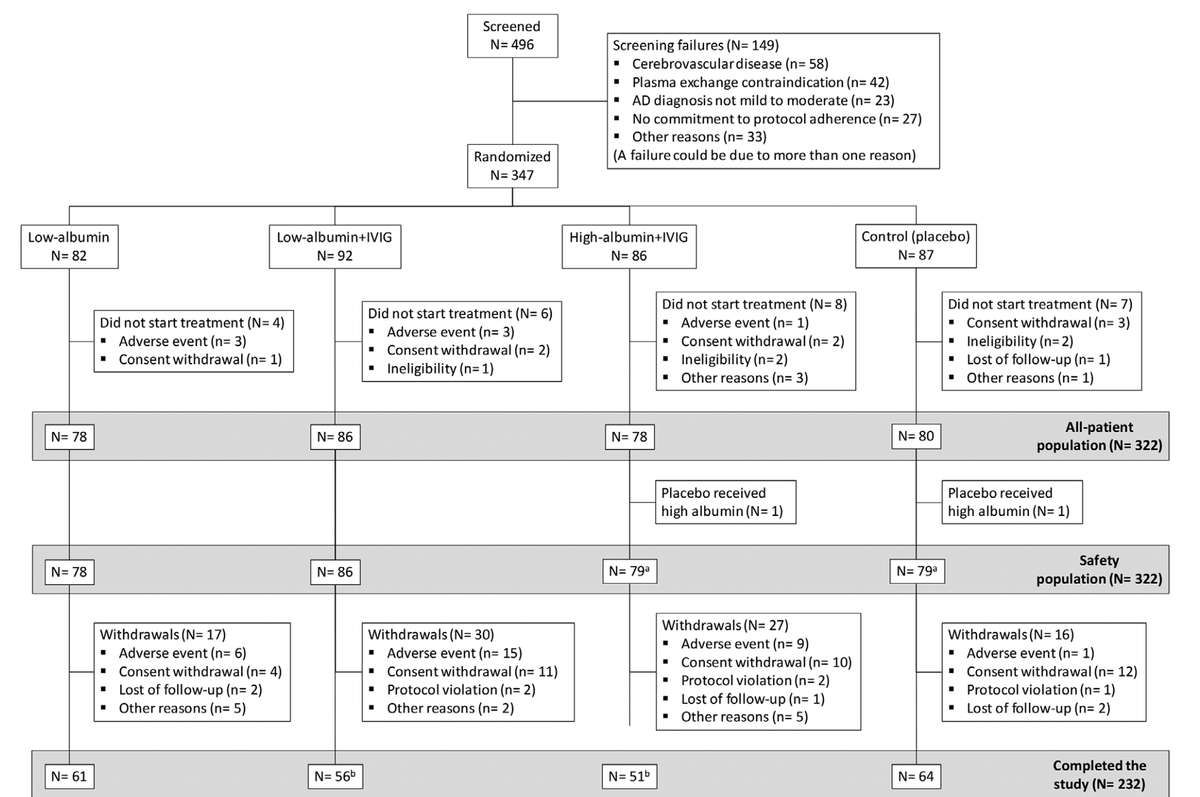
The completion rate ranged from 65-80% depending on treatment arm, which is typical for a 14-month study involving frequent medical procedures.
Treatment Groups
Patients were randomized into four groups:
- Low-albumin group (n=78): 20g albumin infusions during maintenance
- Low-albumin + IVIG group (n=86): 20g albumin alternating with 10g IVIG during maintenance
- High-albumin + IVIG group (n=78): 40g albumin alternating with 20g IVIG during maintenance
- Placebo group (n=80): Sham plasma exchange
The IVIG (intravenous immunoglobulin) was included to counteract potential decreases in endogenous immunoglobulins from repeated plasma removal.
Patient Population
Eligible patients met strict criteria:
- Age 55-85 years
- Diagnosis of probable Alzheimer's disease dementia (NINCDS-ADRDA criteria)
- Mini-Mental State Examination (MMSE) score of 18-26 (mild to moderate disease)
- Stable on acetylcholinesterase inhibitors and/or memantine for at least 3 months
- Brain imaging excluding other causes of dementia
- No severe medical comorbidities
Notably, the study did not require amyloid-positive biomarker status for enrollment. This was based on clinical diagnosis alone, which turned out to be a critical detail we'll discuss later.
What They Measured
The study had two co-primary efficacy endpoints:
1. ADCS-ADL (Alzheimer's Disease Cooperative Study — Activities of Daily Living)
The ADCS-ADL is a 23-item questionnaire scored 0–78, with higher scores indicating better function. Completed by caregivers who know the patient well, it measures real-world abilities like using the telephone, preparing meals, and managing finances. It's the most direct measure of whether a patient can still function independently in daily life.
2. ADAS-Cog (Alzheimer's Disease Assessment Scale — Cognitive Subscale)
The ADAS-Cog is an 11-item test administered directly to patients, scored 0–70 with higher scores indicating worse cognition. It assesses memory, language, orientation, and praxis, and has been the gold-standard cognitive endpoint in Alzheimer's trials for decades.
Secondary endpoints included:
3. CDR-Sum of Boxes (Clinical Dementia Rating — Sum of Boxes)
The CDR-Sum of Boxes evaluates six domains: memory, orientation, judgment and problem-solving, community affairs, home and hobbies, and personal care. Each domain is scored 0–3, giving a total score of 0–18 where higher scores indicate greater impairment. Because it captures such a broad range of functioning, it's arguably the most comprehensive single measure of dementia severity used in the study.
4. ADCS-CGIC (Clinical Global Impression of Change)
The ADCS-CGIC is the blinded clinician's overall assessment of whether the patient has improved, stayed the same, or worsened since baseline — scored on a 7-point scale from marked improvement (1) to marked worsening (7). It integrates all available information about the patient and represents the kind of judgment a physician would make in clinical practice.
5. CSF Biomarkers
- Amyloid-beta 40 and 42 levels
- Total tau and phosphorylated tau proteins
- Measured at baseline, after intensive phase, and after maintenance phase
All clinical measures were assessed at baseline and at months 2, 6, 9, 12, and 14. The primary endpoint was change from baseline to month 14.
Why This Design Matters
This study design is exactly what you want to see in a clinical trial. It was large enough to detect meaningful effects (powered for >90% on primary endpoints), randomized and blinded to prevent bias, conducted across multiple sites to ensure generalizability, and used a sophisticated sham control for a procedural intervention. It included multiple outcome measures across different domains, biomarker assessments to understand mechanism, and long enough follow-up (14 months) to see disease progression.
The fact that dropout rates were similar to other AD trials (20-35%) and that the study was completed as planned speaks to its feasibility.
The Primary Results: What They Found
The moment of truth: Did plasma exchange with albumin replacement actually slow cognitive and functional decline?
Co-Primary Endpoints
When all three treatment groups were pooled together (the "PE-treated" group, n=242) and compared to placebo (n=80), here's what happened:
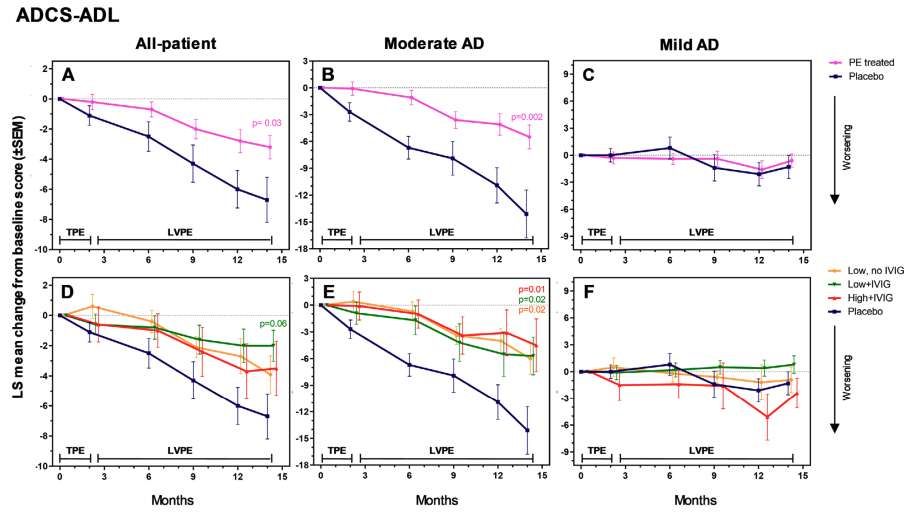
ADCS-ADL (Activities of Daily Living):
The treatment group showed a difference of 3.5 points from placebo (95% CI: 0.4–6.6), a statistically significant result (p=0.03) representing 52% less functional decline.
What this means: Over 14 months, the placebo group declined by about 6.7 points on the ADCS-ADL scale. The treatment group declined by only 3.2 points. That's 52% less functional decline—meaning patients preserved their ability to perform daily activities like managing medications, using appliances, and keeping track of appointments.
ADAS-Cog (Cognitive Function):
The treatment group showed a difference of -2.1 points from placebo (95% CI: -4.4–0.2), a borderline significant result (p=0.06) representing 66% less cognitive decline.
What this means: The placebo group worsened by about 3.2 points on the ADAS-Cog over 14 months. The treatment group worsened by only 1.1 points. While this didn't quite reach the traditional p<0.05 threshold for statistical significance, it showed a strong trend toward benefit, with 66% less cognitive decline.
Understanding P-Values and Effect Sizes
Let me pause here to explain these numbers, because they're crucial to interpreting the results.
P-value: This tells you the probability that the observed difference could have occurred by chance alone. A p-value of 0.03 means there's only a 3% chance this result is due to random variation. Scientists typically use p<0.05 as the threshold for "statistical significance."
The ADAS-Cog result (p=0.06) just barely missed this threshold. Some would dismiss this as "not significant," but in the context of a 322-patient study with consistent effects across multiple measures, a p-value of 0.06 represents strong evidence of benefit. It's more about sample size and statistical power than actual clinical effect.
Effect size: This is arguably more important than the p-value. It tells you the magnitude of the benefit relative to natural disease progression. An effect size of 52% means the treatment group declined only about half as much as the placebo group.
"If we could slow Alzheimer's progression by 50%, we would effectively give patients twice as long at each stage of the disease. That's transformative."
When Did the Effects Appear?
One of the most interesting findings is the timeline of when benefits became apparent. Looking at the graphs of change over time:
Months 0-2 (Intensive Phase): Minimal separation between treatment and placebo groups. Both groups showed similar trajectories.
Months 2-6: The curves began to diverge, with the treatment group showing less decline than placebo.
Months 6-9: Separation became more pronounced, particularly for ADCS-ADL and global measures.
Months 9-14 (Late Maintenance): Maximum separation, with treatment group showing clear benefit across all measures.
This gradual emergence of benefit suggests the mechanism is cumulative rather than immediate. It's not like taking a pill that works within hours. Instead, repeated plasma exchanges seem to have a building effect over 6-9 months.
This timeline is actually encouraging—it suggests that longer treatment might produce even greater benefits. The curves were still diverging at month 14, meaning the full potential effect may not have been captured.
The Secondary Results: Even Stronger Effects
While the co-primary endpoints showed significant or borderline-significant results, the global assessment measures showed even more impressive effects:
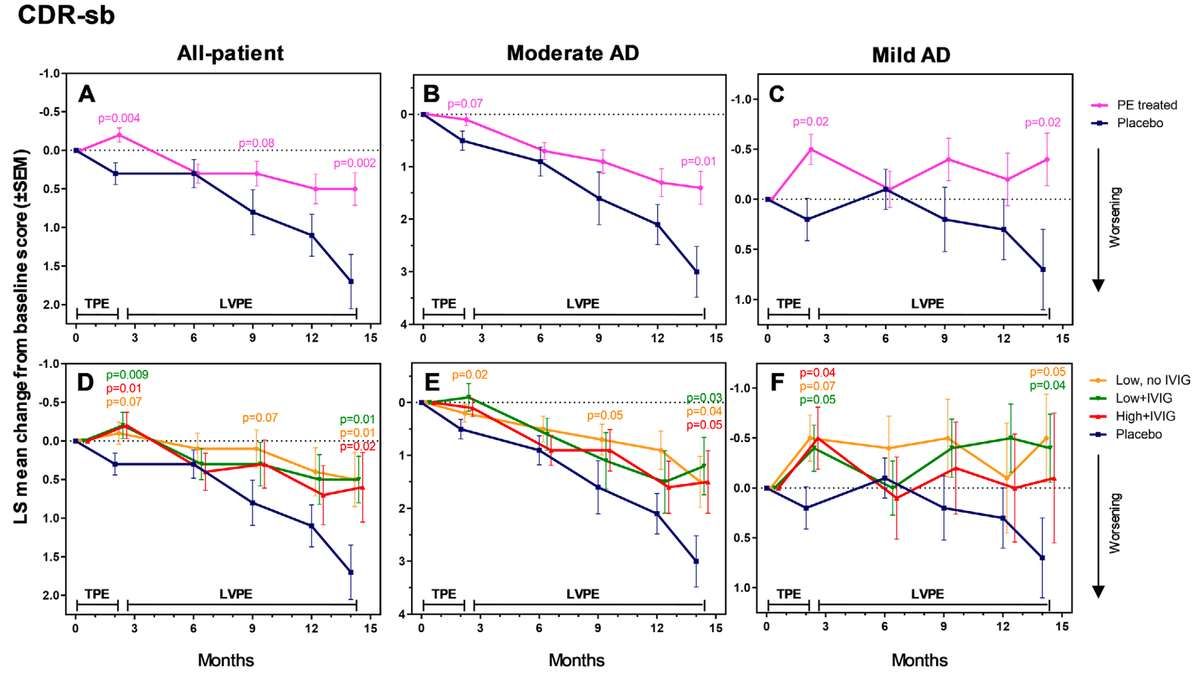
CDR-Sum of Boxes
Across all treated patients, the CDR-Sum of Boxes showed a difference of -1.2 points from placebo (95% CI: -2.2 to -0.2), a highly significant result (p=0.002) representing 71% less overall decline.
The CDR-Sum of Boxes deserves special attention because it's arguably the most comprehensive measure used in the study. Unlike the ADAS-Cog which focuses primarily on cognition, or the ADCS-ADL which focuses on functional tasks, the CDR-sb evaluates six broad domains:
- Memory: Recent and remote memory abilities
- Orientation: Awareness of time, place, and person
- Judgment & Problem Solving: Ability to handle daily decisions and complex situations
- Community Affairs: Managing finances, shopping, business affairs, social functions
- Personal Care: Hygiene, grooming, dressing
Each domain is rated 0-3, and the scores are summed (maximum 18 = severe impairment across all domains).
What makes the CDR-Sb so valuable is that it captures the overall functional impact of dementia on a person's daily life. It reflects how well someone can actually function in their real-world environment—can they manage their finances, maintain their home, care for themselves, and participate in their community?
The 71% effect size on CDR-Sb is remarkable. It was statistically significant at month 14 — and also at earlier time points, with significant separation emerging as early as month 2 (p=0.004) and maintained through the end of the study (p=0.002).
This consistency across time points strengthens confidence that this is a real effect, not a statistical fluke.
ADCS-CGIC (Clinical Global Impression of Change)
The ADCS-CGIC showed a difference of -0.8 to -0.9 points from placebo (95% CI: -1.2 to -0.4), an extremely significant result (p<0.0001) representing effective stabilization versus decline in the placebo group — an effect size of 100–113%.
The ADCS-CGIC is the clinician's overall judgment of whether the patient has improved, stayed the same, or worsened since baseline. It's scored on a 7-point scale:
- 1 = Marked improvement
- 4 = No change
- 7 = Marked worsening
In the placebo group, the average score was around 4.8, indicating patients had worsened. In the treatment groups, the average score was around 4.0, very close to "no change."
An effect size of 100% means the treatment essentially prevented decline rather than just slowing it. This is what we see with the ADCS-CGIC—treated patients remained stable while placebo patients declined.
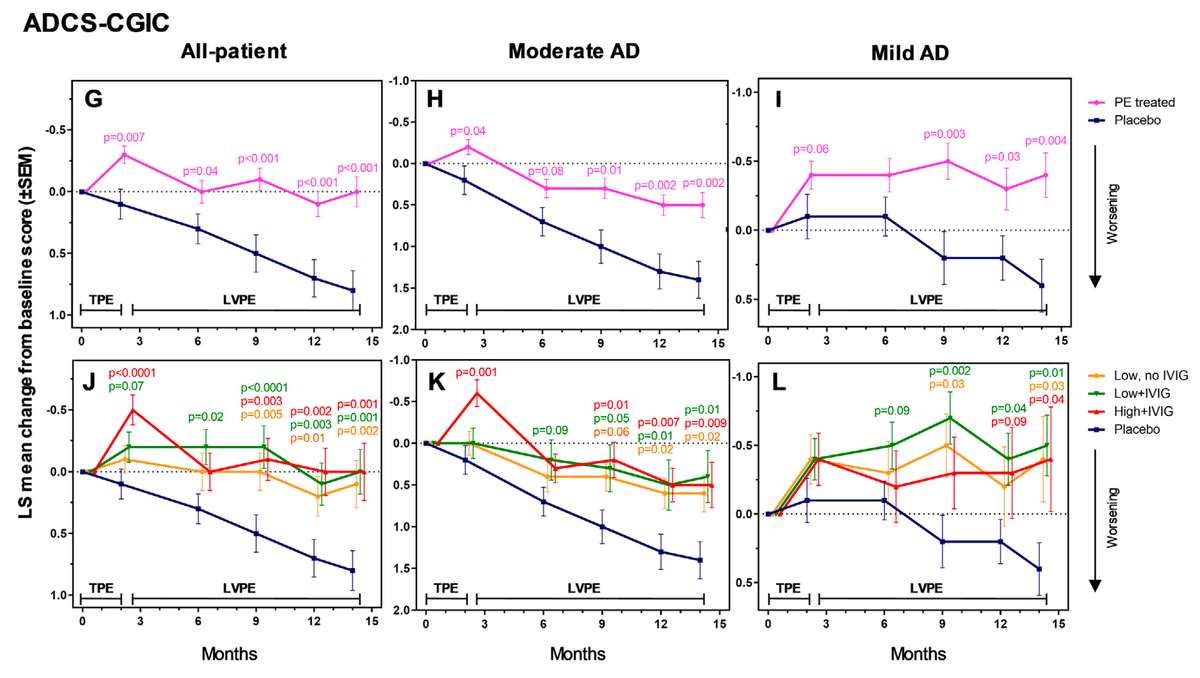
The fact that this global clinical assessment showed such strong effects (p<0.0001 is about as certain as you can get in a clinical trial) suggests that experienced clinicians, blinded to treatment assignment, could clearly see a difference between treated and placebo patients.
The Subgroup Analysis: Disease Severity Matters
The treatment worked differently in mild vs. moderate Alzheimer's—with moderate patients showing 61% less decline across all measures, while mild patients actually improved on some tests.
One of the most important findings from AMBAR came from pre-specified subgroup analyses based on baseline disease severity. Patients were divided into two groups based on their baseline MMSE scores:
- Mild AD: MMSE 22-26 (n=161)
- Moderate AD: MMSE 18-21 (n=161)
The results in these subgroups revealed dramatically different patterns.
Moderate AD Patients: Clear Slowing of Decline
In patients with moderate Alzheimer's (MMSE 18–21), the effects were strongest and most consistent. The ADCS-ADL showed a difference of 8.6 points (p=0.002), representing 61% less functional decline. The ADAS-Cog showed a difference of -3.9 points (p=0.05). The CDR-Sum of Boxes showed a difference of -1.5 to -1.8 points (p=0.01 to 0.04), representing 50–60% less overall decline. And the ADCS-CGIC showed a difference of -0.8 to -1.0 points (p=0.002 to 0.02), representing 57–71% less decline on clinician global impression. All three individual treatment arms showed statistically significant benefits on the ADCS-ADL when analyzed separately — a consistency across dosing regimens that strengthens the finding. The pattern is clear: plasma exchange slowed progression across cognitive, functional, and global measures in moderate AD patients.
Mild AD Patients: A Different Story
In patients with mild Alzheimer's (MMSE 22–26), the results told a different story. Standard cognitive measures like the ADAS-Cog showed no significant changes in either direction — likely a ceiling effect, since these tests aren't sensitive enough to detect subtle changes in people with milder impairment. But the global functional measures revealed something unexpected. On the CDR-Sum of Boxes — where lower scores indicate less impairment, and higher scores indicate more — treated patients actually improved by 0.8 to 1.2 points from baseline while the placebo group worsened (p=0.02 to 0.04). The same pattern held on the ADCS-CGIC — where a score of 4 means no change and higher scores mean worsening — with treated patients scoring 0.8 points better than placebo (p=0.004 to 0.04). Effect sizes of 157–200% reflect the fact that the treatment group didn't just decline less than placebo — they moved in the opposite direction entirely. In a progressive neurodegenerative disease, that's not supposed to happen.
"In a progressive neurodegenerative disease, we don't expect to see improvement. Yet mild AD patients who received plasma exchange showed slight improvement over 14 months on global functional measures."
What This Tells Us
The different patterns in mild versus moderate AD are revealing.
In moderate AD: Plasma exchange slowed decline across all measures. The effect was to preserve function and cognition that would otherwise have been lost.
In mild AD: The disease-specific cognitive tests (ADAS-Cog) weren't sensitive enough to show change in either group. But the broader functional measures (CDR-sb, ADCS-CGIC) showed actual improvement in treated patients.
This suggests that plasma exchange may help restore or enhance certain aspects of functioning, not just slow their loss. This is more consistent with a treatment that addresses multiple pathological processes (inflammation, oxidative stress, vascular dysfunction) rather than one that simply removes a single toxic protein.
Read the Complete Series:
